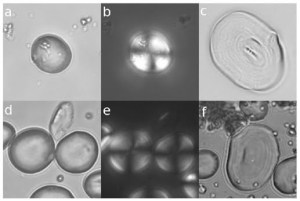
Microscopic barley grains. Top row are examples of grains from Shanidar calculus, and beneath each are examples of modern barley to which they are probably related. Fig. 1 from Henry et al. (in press)
Author: zcofran
Unwarranted zeal: Melvin Moss on modern methods
I’m doing some reading on the study of craniofacial growth, I stumbled across this poignant quote from Melvin Moss, from a seminar on “New Techniques in Processing and Handling Growth Data”:
“This is very beautiful. It is neat, it is modern technology, and it is fast. I am just wondering very seriously about the biological validity of what we are doing with this machine.” (Moyers & Krogman, eds: p. 326)
Atavisms: talk about old school
This month’s Current Biology has a “Quick Guide” segment by Brian Hall on atavisms: the occasional and random appearance of ancestral traits in individuals of species that no longer have that trait. Examples Hall provides are vestigial hindlimbs (legs or fins) occasionally found on dolphins or snakes, which evolved from animals that did have limbs.
Growing a Homo erectus kid, sort of
A paper, given at this year’s Physical Anthropology meetings, was just published online in the Journal of Human Evolution, with a re-evaluation of the height and possible growth pattern of a subadult skeleton of Homo erectus (KNM-WT 15000, aka “Nariokotome boy,” aka “Stripling youth”). When initially described, it was estimated that this young chap would gave grown to be around 6 feet tall. However, controversy around the skeleton’s age at death and probable growth pattern have made this quite a contentious topic. In the recent paper, Ronda Graves and colleagues used a South African human growth pattern and a pattern from “naturally-reared captive” chimpanzees to devise a series of intermediate growth patterns that might have characterized H. erectus. Using the pattern they felt most likely reflected the Nariokotome skeleton’s estimated life history parameters, the authors estimate the potential adult height of the youth to have been closer to about 5′ 4″.
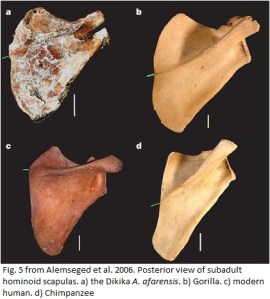
"Big Man" and the scapula of Australopithecus afarensis
 Last November I reported on recently described Australopithecus cf. afarensis craniodental remains from the site of Woranso Mille in Ethiopia. These fossils are significant in part because they date to around 3.6 million years ago; most of the postcranial evidence for A. afarensis comes from Hadar (~3.4 – 2.9 million years) or Maka (~3.5 million years). It is pretty awesome, then, that Yohannes Haile-Selassie and colleagues (2010a) have just reported on a partial skeleton from Woranso-Mille.
Last November I reported on recently described Australopithecus cf. afarensis craniodental remains from the site of Woranso Mille in Ethiopia. These fossils are significant in part because they date to around 3.6 million years ago; most of the postcranial evidence for A. afarensis comes from Hadar (~3.4 – 2.9 million years) or Maka (~3.5 million years). It is pretty awesome, then, that Yohannes Haile-Selassie and colleagues (2010a) have just reported on a partial skeleton from Woranso-Mille.

Evolution of human fingers and toes: The two go foot in hand
A really cool study was just published in the journal Evolution, and what with getting my apartment ready for a New Year’s party on the 31st, and my being completely incapacitated yesterday, I didn’t get to read through it until today. Campbell Rolian and colleagues (in press) address the question: In human evolution, were hand and foot digital proportions each the targets of direct selection, or could hand/foot proportions have evolved as a byproduct of selection on only the hand or only the foot?
This is an interesting question. In your standard Anthropology 101 class, you learn about how humans (and hominins) are unique relative to apes. Two unique things about us are: a robust, adducted big toe for bipedalsim, and a hand adapted for tasks requiring a fairly high degree of dexterity, such as tool use. But something to keep in mind–indeed the authors of this study did–is that the hand and foot are serially homologous, each is a variant on a common theme. Because the developmental architecture behind the hand and foot are largely similar, an intuitive question is whether selection on the hand or foot only would effect the evolution of the element that wasn’t under selection. Could developmental integration of the hominin hand and foot have led to evolutionary integration, do/did the hand and foot co-evolve?
Turns out this may well be the case. Authors looked at lengths and widths of hand and foot phalanges (finger bones) in a sample of humans and chimpanzees. Generally, in both Pan and Homo, homologous traits in the hand and foot are more highly correlated than expected by chance, even compared to correlations between traits within the hand and foot. Cool!
But then the authors did some crazy simulations, to see what kinds of selection regimes on the hand and foot may have led from a chimp-like morphology to the morphology we humans enjoy today. I’ll need to reread this section a couple times, but it looks like selection on the big toe is one of the most important aspects of hominin hand/foot evolution. And it would not be impossible for evolutionary changes in the human hand to be largely by-products of selection on the foot, due to the nature of covariation (integration) of the hand and foot. Whoa!
The implication, which the authors seem to like, is this: given a chimp-like ancestral morphology for the hand and foot, it seems that the two major hominin/human traits given above (bipedalism and tool-use/manual dexterity) are largely due to selection simply on the foot. That is, because of the developmental integration of the hand and foot, selection for a bipedally capable foot indirectly induced the evolution of a hand conducive to manipulation. Ha, the hand was just along for the ride! Get it, because the feet move the body, and so the hand… but also evolutionarily… Dammit.
Anyway, that’s nuts! Of course, another very interesting thing about the first digits of the human hand and foot, aside from the fact that the first digit on both is relatively large and robust, is that the mobility of these digits is just about opposite what it is in the apes. Whereas the big toe is very mobile/opposable in apes (and the 4.4 million year hold putative hominin, Ardipithecus ramidus), it is completely adducted in humans (and fossil hominins that aren’t Ar. ramidus). Less extreme, the human thumb joint is allegedly more mobile than apes’ thumbs. So this is the next step, I guess: what is the developmental basis for the wild evolution of the human hallux and pollex joints?
Reference
Rolian C, Lieberman DE, and Hallgrimsson B. Coevolution of human hands and feet. Evolution: in press.
Pongo amidst conservation and industry
The December issue of Current Biology has a short summary about collaborations between the palm oil industry and conservationists to preserve orangutan (Pongo pygmaeus) habitats in Borneo. As the palm oil industry has burgeoned, orangutan populations have lost contact due to deforestation for industry and agriculture. Apparently palm oil companies have made an agreement with the government of the Malaysian state of Sabah in Borneo, in which the companies will help construct corridors that will reconnect isolated populations of the orangutan.
 Lethargic orangutan at the Zoo Atlanta, in Hottlanta GA. He was cool but boring because he didn’t do anything.
Lethargic orangutan at the Zoo Atlanta, in Hottlanta GA. He was cool but boring because he didn’t do anything.Good to see cooperation rather than conflict between conservationists and industries. Let’s hope it proves beneficial for the endangered orangutans.
Assuming the project works out, it will be interesting to see population genetics and behavioral studies documenting the results of renewed contact and gene flow of these erstwhile isolated apes. Since the prior isolation and future reconnection are anthropogenic, or due to human activity, it will be an interesting (and hopefully not too depression) lesson about how human behavior affects biodiversity.
On an aside, I just heard, “Goonies never say ‘die'” (Sean Astin, Goonies).
Reference
Williams N. 2009. Orang-utan plan. Current Biology 19: R1098
Hybrids, hominoids and hominins
Is the Eastern lowland gorilla (Gorilla beringei graueri) a hybrid (sub)species? A recent study by RR Ackermann and JM Bishop suggests this scenario.
Their study used morphological, genetic and geographic information to analyze variation in extant gorilla species and subspecies. A previous study by Ackermann and colleagues (2006) on baboons found that non-metric traits–namely pairs of extra teeth and unusual sutures between some facial bones–have freakishly high frequencies in known hybrids compared to their parents of different species. Well wouldn’t you know it: G. b. graueri had a significantly higher frequency of such traits than the other gorilla species/subspecies (the putative ‘parental’ species, the Eastern mountain gorilla G. b. beringei and the Western lowland gorilla G. gorilla gorilla).
Additionally, for a number of cranial metric traits, graueri had significantly higher values than the other gorilla species’ sample averages. “Heterosis” refers to a condition wherein a hybrid phenotype exceeds the combined parental mean–it appears that if this is truly a hybrid population, graueri displays heterosis for a number of cranial features. Finally, it is notable that graueri has been described as more like Western gorillas in some respects, but more like Eastern mountain gorillas in others, and then totally unique in some aspects. This is arguably a result of graueri possessing genes from two other distinct species.
Oh, and the mtDNA evidence suggests fairly recent gene flow from Western lowland gorillas eastward. Because mtDNA is maternally inherited, this implies that females have been involved in this west-to-east gene flow. However, it is unclear the extent to which there was east-to-west gene flow, or the potential involvement of male gorillas here.
Why is graueri likely a largely hybrid sample, and not just part of a morphological and genetic cline conecting Western and Eastern gorilla populations (i.e. making gorillas a single, polymorphic, geographically broad species)? The hybrid morphologies above are believed to indicate complications that arise in development, due to the union of two species’ distinct sets of genes. Such signatures of hybridizaiton would not be expected to appear in a regularly panmictic species. It also seems that the separation of Western and Eastern gorilla species occurred during the Pleistocene, which means that the two sides have been diverging for quite a long time and have recently come back into contact.
I think the authors make a very good case for the importance of hybridization in the evoluton of gorillas, at least as we know them today. I like their use of both morphological and genetic data, which complement one another nicely in support of a hybrid-type nature of Gorilla beringei graueri. In addition, the implicaitons of the study are fantastic! Even though the authors did not know for sure whether individual specimens were hybrids, they were able to use the results of previous work to make a convicning case that a number of their specimens were very likely to be hybrids. This is a good sign for persons like myself who are interested in the detection of hybrids in skeletal/fossil samples. Another great implication is that hybridization indeed has a place in hominoid evolution–it awaits to be seen what role hybridization may have played in the course of human evolution.
References
Ackermann RR, Rogers J and Cheverud JM. 2006. Identifying the morphological signatures of hybridization in primate and human evolution. Journal of Human Evolution 51: 621-645.
Ackermann RR and JM Bishop. Morphological and molecular evidence reveals recent hybridization between gorilla taxa. Evolution: in press.
Bridging the gap: Australopithecus from Woranso
Recently discovered Australopithecus fossils from the Ethiopian site of Woranso-Mille help fill a gap between parts of the early hominin fossil record (Haile-Selassie et al, in press). The fossils date to between 3.8-3.6 million years ago (Ma), and consist of several teeth and a jaw fragment. These specimens show a number of features that are intermediate in morphology between the earlier Au. anamensis (4.2-3.9 Ma) and later Au. afarensis from Laetoli (~3.7-3.5 Ma). As a result, the Woranso fossils lend support to the hypothesis that Au. anamensis and Au. afarensis represent a single evolving species (i.e. Kimbel et al. 2006).
I think this is exciting for two reasons. First, the fossils bridge the morphological gap between the older anamensis and younger afarensis fossils. As a result, we get to ‘see’ anagenetic evolution—changes within a single lineage. One topic in evolutionary biology is about the mode and tempo of evolution: are species fairly constant, then evolve into multiple ‘daughter’ species (“punctuated equilibrium”); or does evolutionary change tend to occur more within individual lineages (“anagenesis”)? Obviously neither is mutually exclusive, rather evolution is probably best characterized variously by both processes. Still, in the world of paleoanthropology, where many researchers argue for rapid and constant species turnover within the human lineage, it is cool to see a convincing argument for anagenesis. However, this ignores the meager (but intriguing) K. platyops material (Leakey et al. 2001), dating to around 3.5 Ma, possibly indicating the proliferation of at least two hominin species shortly after 4 Ma.
Second, the morphological intermediacy of the Woranso fossils allow a look at the patterns of evolutionary change within the anamensis–afarensis lineage. The authors note that the teeth of the Woranso hominins are generally more similar to anamensis, but have some derived characters of the later afarensis teeth. If we truly have a glimpse of dental evolution within a single lineage, we can ask questions about the evolution and development (“Evo-Devo”) of teeth. Are changes in these teeth correlated in a way that could be predicted by certain developmental models? Or is selection acting independently on various tooth traits?
References
Haile-Selassie Y, Saylor BZ, Deino A, Alene M, and Latimer BM. New hominid fossils from Woranso-Mille (Central Afar, Ethiopia) and Taxonomy of Early Australopithecus. American Journal of Physical Anthropology, in press.
Kimbel WH, Lockwood CA, Ward CV, Leakey MG, Rak Y, and Johanson DC. 2006. Was Australopithecus anamensis ancestral to A. afarensis? A case of anagenesis in the hominin fossil record. Journal of Human Evolution 51: 134-152.
Leakey MG, Spoor F, Brown FH, Gathogo PN, Kiarie C, Leakey LN, and McDougall I. 2001. New hominin genus from eastern Africa shows diverse middle Pliocene lineages. Nature 410: 433-440.
Claude Levi-Strauss: 1908-2009
The great, dare we say legendary, anthropologist Claude Levi-Strauss passed away this past weekend at the ripe age of 100. Levi-Strauss is largely responsible for establishing the structuralist school in anthropology, which emphasized the prevalence of binary opposition in culture, myths, language, and the like. I recall reading some of his work for a Theories of Myth course, which provided a fascinating of viewing the creation and roles of myths. Few anthropologists have had such an impact within their own field and beyond, as Claude Levi-Strauss.