Update: research in this post was eventually published in American Journal of Primatology here
I’m back in Astana, overcoming jet lag, after the annual conference of the American Association of Physical Anthropologists, which was held in my home state of Missouri. I’d forgotten how popular ranch dressing and shredded cheese is out there. It was also nice to be surrounded by colleagues interested in evolution, primates, and fossils.
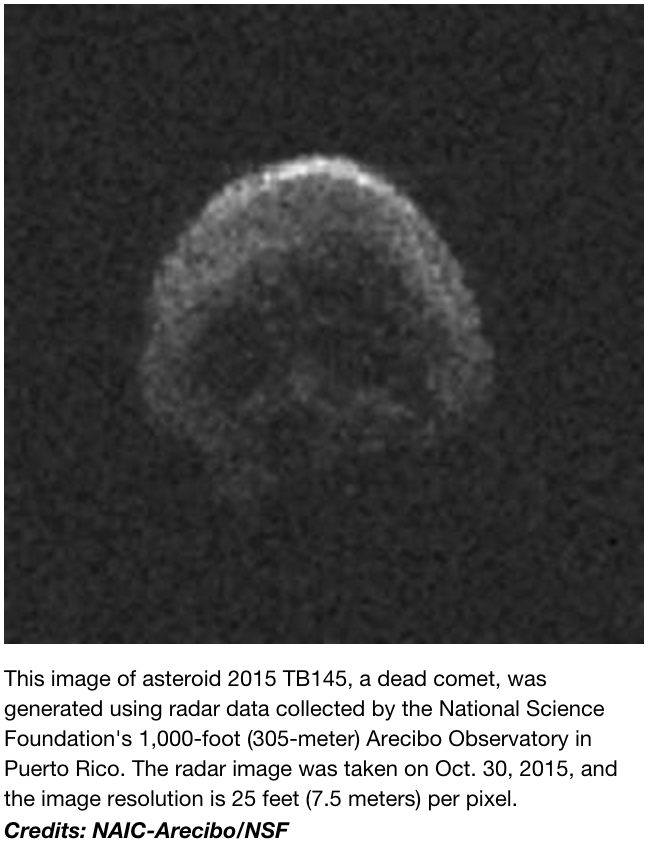
Although I usually present in evolution and fossil-focused sessions, my recent interest in brain growth landed me in a session devoted to Primate Life History this year. The publication of endocranial volumes (ECVs) from wild chimpanzees of known age from Taï Forest (Neubauer et al., 2012) led me to ask whether this cross-sectional sample displays the same pattern of size change as seen in captive chimpanzee brain masses (Herndon et al., 1999). These are unique datasets because precise ages are known for each individual, and this information is generally lacking for most skeletal populations. We therefore have a unique opportunity to estimate patterns and rates of growth, and to compare different populations. Here are the data up to age 25 (the oldest known age of the wild chimps):

Brain size plotted against age in chimpanzees. Blue Ys are the Yerkes (captive) apes and green Ts are the Taï (wild) chimps. Note that Yerkes data are brain masses while the Taï data are endocranial volumes (ECVs). Mass and volume – as different as apples and oranges, or as oranges and tangerines? Note the relatively high “Y” at 1.25 years, who was omitted from the subsequent analysis.
This is an interesting comparison for a few reasons. First, to the best of my knowledge brain size growth hasn’t been compared between chimp populations (although it has been compared between chimps and bonobos: Durrleman et al., 2012). Second, many studies have found differences in tooth eruption, maturation and skeletal growth and development between wild and captive animals, but again I don’t think this has been examined for brain growth. Finally, and most fundamentally, it’s not clear whether ECV and brain mass follow the same basic pattern of change (brain mass but not ECV is known to decrease at older ages in humans and chimps, but at younger ages…?.
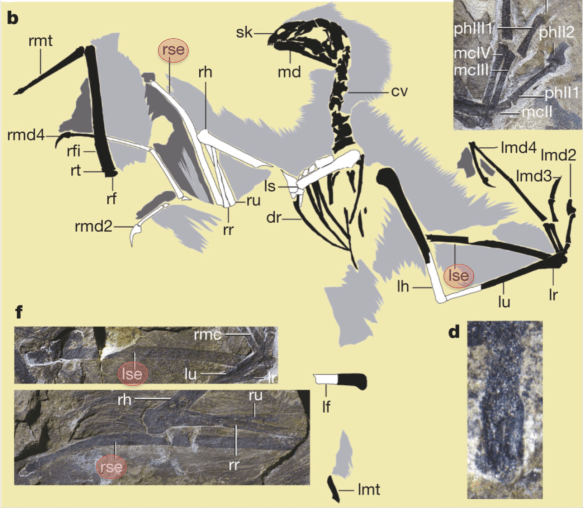
So to first make the datasets comparable, I used published data to examine the relationship between brain mass and ECV in primates, to estimate the likely ECV of the Yerkes brain masses. Two datasets examine adult brain size across different primate species (red and blue in the plot below), and one looks at brain mass and ECV of individuals for a combined sample of gorillas (McFarlin et al., 2013) and seals (Eisert et al., 2013). In short, ECV and brain mass in these datasets give regression slopes not significantly different from 1. One dataset has a negative y-intercept significantly different from 0, meaning that ECV should actually be slightly less than brain mass, but I think this pattern is driven by the really small-brained animals like New World Monkeys).

The relationship between endocranial volume and brain mass in primates (and Weddell seals). Solid lines and shaded confidence intervals are given for each regression, and the dashed line represents isometry, or a 1:1 relationship (ECV=brain mass). The rug at the bottom shows the range of the Yerkes masses. Note that the red and black regressions are not significantly different from isometry, while the blue regression is shifted slightly below isometry.
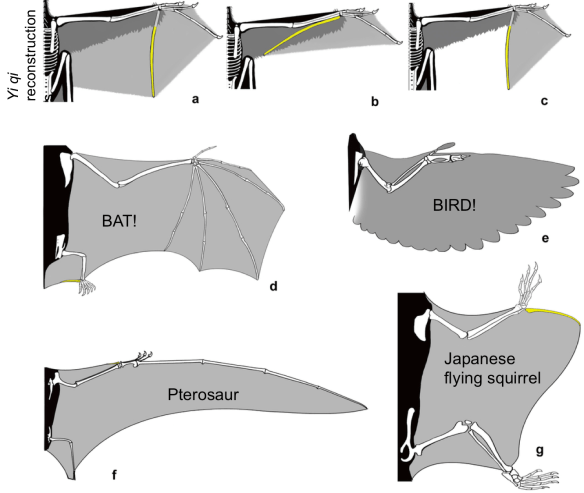
So let’s assume for now that the ECVs of the Yerkes apes are the same as their masses, meaning the two datasets are directly comparable. There are lots of ways to mathematically model growth, and as George Box famously quipped, “All models are wrong, but some are useful.” Here, I wanted to use something that explained the greatest amount of ontogenetic variation in ECV while also levelling off once adult brain size was reached (by 5 years based on visual inspection of the first plot above). This led me to the B-spline. With some tinkering I found that having two knots, one between each 0.1-2.5 and 2.6-5, provided models that fit the data pretty well, and I resampled knot combinations to find the best fit for each dataset. The result:

B-splines describing the relationship between ECV (or brain mass) and age in the TaÏ (green) and Yerkes (blue) data. Note that although the Yerkes line is elevated above the Taï line after 4 years, the confidence intervals (shaded regions) overlap at all ages.
These models fit the data pretty well (r-squared >0.90), and nicely capture the major changes in growth rates. Resampling knot positions reveals best-fit models with different knots for each sample, but otherwise the two models cannot be statistically distinguished from one another: the 95% confidence intervals of both the model coefficients and brain size estimates overlap. So statistical modelling of brain growth in these samples suggests they’re the same, but there are some hints of difference.

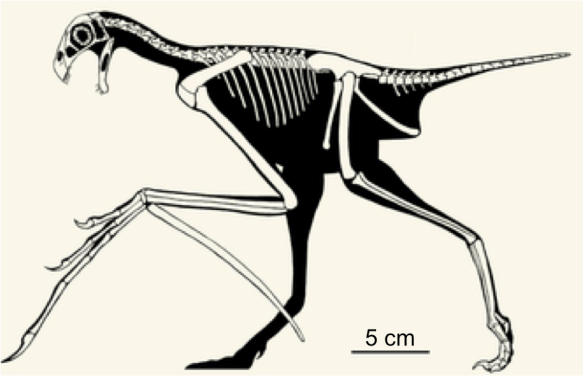
Growth rates at each age calculated from the B-spline regressions. Note these are arithmetic velocities and not first derivatives of the growth curves. The dashed horizontal line at 0 indicates the end of brain size growth.
Converting the growth curves to arithmetic velocities we see what accounts for the subtle differences between samples. The velocity plot hints that, in these cross-sectional data, brain size increases rapidly after birth but growth slows down and ends sooner in Taï than among the Yerkes apes. I’m cautious about over-interpreting this difference, since there is great overlap between growth curves, and there is only one Taï newborn compared to about 20 in Yerkes: even just a few more newborns from Taï might reveal greater similarity with Yerkes.
So there you have it, it looks like the wild Taï and captive Yerkes chimps follow basically the same pattern of brain growth, despite living in different environments. Whereas the generally greater stressors in the wild often lead to different patterns of skeletal and dental development in wild vs. captive settings, brain growth appears pretty robust to these environmental differences. That brain growth should be canalized is not too surprising, given the importance of having a well-developed brain for survival and reproduction. But it’s cool to see this theoretical expectation borne out with empirical observations.