The Fall semester here at Vassar kicks off next week, and so of course a new fossil discovery is published this week that threatens to upend my course plans and throw my syllabi into disarray. Haile-Selassie and colleagues report a very well-preserved hominin cranium, from the Woranso-Mille region of Ethiopia and dating to 3.8 million years ago. The new cranium shares features with Australopithecus anamensis, a species previously mainly known through jaws and teeth. The fossil is therefore really important since it puts a face to the species’ name, and it is the oldest relatively complete Australopithecus cranium known. When I showed a picture of the fossil to my wife, who is not a paleoanthropologist, all she said was that it looked like the face of a dog who got stung by a bee.

The new A. anamensis fossil MRD-VP-1 (left), and a dog that lost a fight with a bee. Fossil photo from the Smithsonian‘s coverage.
The big buzz in many news stories about the fossil (for example, Nature, ScienceNews, etc.) is that it rewrites an evolutionary relationship early in human history, with Australopithecus anamensis no longer the ancestor of A. afarensis, but rather the two being contemporaries. That idea is based on a 3.9 million year old frontal bone attributed to A. afarensis from a site called Belohdelie, also in Ethiopia (Asfaw, 1987): basically, the new A. anamensis cranium reveals a hominin with a narrow frontal region of the brain, which lived 100,000 later than A. afarensis with a relatively expanded frontal region:
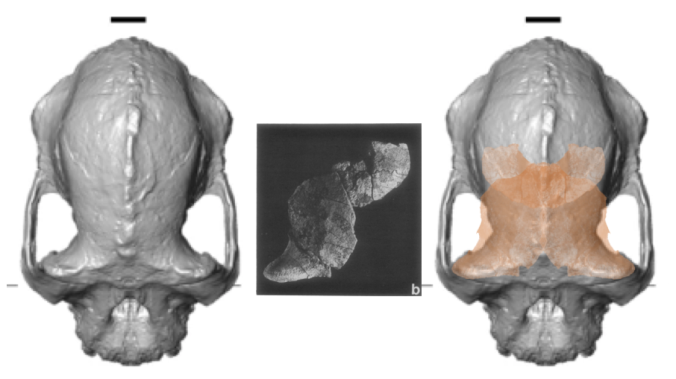
Top views of the reconstructed A. anamensis cranium (left), and the Belohdelie frontal (center), and my crappy photoshopped overlay of Belohdelie on A. anamensis (right). Images not to scale.
The lede, “human evolutionary tree messier than thought,” is not terribly interesting or compelling since it seems to characterize most fossil discoveries over the past several years. And in this case I don’t know how well supported the argument is, since the trait in question (narrow frontal region of the braincase or “post-orbital constriction”) can vary dramatically within a single species. The image below is from the paper itself—compare the difference in “postorbital constriction index” (left graph) between the new A. anamensis cranium (MRD) and A. afarensis (in blue). Both sets of fossils fall within the range of chimpanzees (P. troglodytes), and note the great range of variation within gorillas (G. gorilla).

Part of Figure 3 from the paper by Haile-Selassie and colleagues. On the top is a view from above of fossil humans: Sahelanthropus tchadensis, Ardipithecus ramidus, the new A. anamensis, A. afarensis, and A. africanus. Below the graphs show how species differ in narrowing of the frontal (left) and length of the skull (right).
What I find most interesting about the new find is the great front-to-back length of the cranium—check out how long and narrow the brain-case is of the fossil compared with the later hominins to the right. This is an interesting similarity with the much earlier (6 million years ago) Sahelanthropus tchadensis, which is the left-most fossil in the figure. It makes me really curious to see the brain endocast of A. anamensis and the Sahelanthropus cranium—what was brain shape like for these ancient animals, and what does that mean for the earliest stages of human brain evolution? The Sahelanthropus endocast was meticulously reconstructed and analyzed by Thibaut Bienvenu (dissertation here), who found it to be somewhat like gorillas in overall shape (but with a more hominin-like placement of the foramen magnum). Of course, the outside of the skull doesn’t necessarily tell us what the underlying brain shape was like. Haile-Selassie and colleagues report that they made a virtual reconstruction of the A. anamensis endocast, so hopefully we’ll get to pick its brain soon.