The great, dare we say legendary, anthropologist Claude Levi-Strauss passed away this past weekend at the ripe age of 100. Levi-Strauss is largely responsible for establishing the structuralist school in anthropology, which emphasized the prevalence of binary opposition in culture, myths, language, and the like. I recall reading some of his work for a Theories of Myth course, which provided a fascinating of viewing the creation and roles of myths. Few anthropologists have had such an impact within their own field and beyond, as Claude Levi-Strauss.
Author: zcofran
Halloween special: Heterotopy, pleiotropy, and the origins of vampires (free dissertation topic/movie plot-line)
 In the spirit of Halloween, I’d like to propose a dissertation topic. Vampires are all the rage now, for a reason I just don’t understand. Seeing a popular portrayal of these monsters (an episode of HBO’s True Blood, see figure to the right), I noticed that the vampires’ fangs are in the position of their lateral incisors, not their canines. This is odd, since after all, in humans, hominins and most other primates, the canine teeth are the relatively sharp ones–what gives?
In the spirit of Halloween, I’d like to propose a dissertation topic. Vampires are all the rage now, for a reason I just don’t understand. Seeing a popular portrayal of these monsters (an episode of HBO’s True Blood, see figure to the right), I noticed that the vampires’ fangs are in the position of their lateral incisors, not their canines. This is odd, since after all, in humans, hominins and most other primates, the canine teeth are the relatively sharp ones–what gives?
At first I thought it had to do with Hollywood logistics–putting the fangs in the canine position would be too far over for them to be easily visible normally. Putting them in the lateral incisor position, the fangs are now easily visible at all times. But wait! Could there be another, more complex answer?
What if, now stay with me on this, what if it turns out that this isn’t just Hollywood–what if this is an example of heterotopy (development of a feature in a novel location). That is, something about vampirism also causes the canine to develop in the the position of the lateral incisor. Could this be due to pleiotropy? That is, perhaps genes underlying vampirism are also involved in dental development, or vice versa. Could the pleiotropic effects of this heterotopic change in canine development result in vampiric symptoms like immortality, bloodlust, and garlic aversion? Or could selection for vampirism (bloodlust?) have the added (pleiotropic) effects of disdain for garlic and crucifixes, and a heterotopic shift in canine development?
So here’s a free dissertation topic: untangling the genetic architecture of vampirism. I’d do it, but I have other, actual things to work on. Incidentally, if anyone wants to use this idea for a book or movie, please ask first. I know you’re all beating down my door for this one.
Convergent Evolution and Primate Origins
Hooray, the third post in a short amount of time about convergent evolution! Just found a paper in press in Journal of Human Evolution, where authors demonstrate that the convergent evolution in primates and some birds suggests that the earliest primates initially filled a visual-predator niche, and that the bony covering surrounding the haplorhine primate (tarsiers, monkeys, apes, and people) eyes serves to protect the eyes from the adjacent chewing muscles.
The authors note that one hallmark of primates are their forward-facing, convergent orbits (fancy name for where the eyes sit in the skull). Back in the 1970s Matt Cartmill suggested that this helped increase the visual acuity of the earliest primates, who used their sharp sense of vision to hunt for small insects, probably at night. So to test such a “nocturnal visual predation hypothesis,” they compared the orbits of 103 species of birds. Lo and behold, strigiform birds (you may know them as owls, I know them as delicious), which are visual predators and mostly nocturnal, have the most orbital convergence.
In addition, haplorhines are unique among primates in having a “post-orbital septum,” or a bony plate separating the orbit from the temporal fossa behind it (though the tarsier’s septum is incomplete). Just as haplorhines are unique among primates, so too are owls among birds, in having a large, bony projection behind the eye. It has been suggested that this morphology serves to keep the temporalis muscle from pushing against the eye during chewing. To test this hypothesis regarding the function of bony protection behind the eye, the authors dissected a few owls. As predicted, the dissection showed the owls’ projection deflects the path of the adjacent jaw muscle.
I think this study is neat for two reasons: first, it’s another great example of the pervasiveness of convergence in evolution. In spite of how diverse animal forms are, they often have very similar features, which makes it likely that diverse animals will evolve similar traits or behaviors in similar ways, under selection. If the ancestral condition in birds and pre-primates was to have eyes that don’t face forward, and there was selection to increase visual acuity for predation, then it looks like each group had no evolutoinary ‘choice’ but to move their eyes forward.
Second, the study demonstrates the utility of the comparative method to test functional hypotheses. While all primates have forward-facing eyes, modern primates are so diverse that it is hard to say why exactly such a feature evolved in the first place. By seeing why the feature evolved in certain birds, we can infer why it evolved in our earliest ancestors.
Reference
Menegaz R and Kirk EC. Septa and processes: Convergent evolution of the orbit in haplorhine primates and strigiform birds. Journal of Human Evolution, in press.
What do you call a Middle Miocene hominoid that doesn’t belong to you?
Nacholapithecus kerioi.
Oh man, that was a bad joke. N. kerioi is an ape known from the site of Nachola in Kenya, dating to 17-14 million years ago. There is a fairly complete skeleton, KNM-BG 35250, and completeness is always exciting for the paleontologist. It seems to have had a fairly large and robust forelimb compared to its hindlimb, possibly indicating locomotor behavior unlike anything modern primate. Based on evidence from the skull and teeth it was likely a hard-object feeder, a characteristic in many Miocene hominoids starting with the 17 million year-old Afropithecus. Here’s a picture of the skeleton (Nakatsukasa and Kunimatsu 2009, Fig. 1):
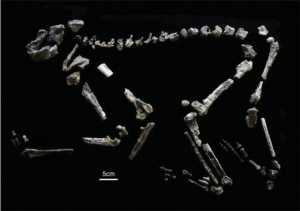 This specimen is the holotype for N. kerioi. A nice contrast to my last post griping about specimens that have been selected as holotypes.
This specimen is the holotype for N. kerioi. A nice contrast to my last post griping about specimens that have been selected as holotypes.
Reference
Nakatsukasa M and Kunimatsu Y. 2009. Nacholapithecus and its importance for understanding hominoid evolution. Evolutionary Anthropology 18: 103-119.
Australopithecus afarensis: A mythical creation of Jim Henson?
DIK-1-1 is a nearly complete juvenile Australopithecus afarensis skeleton, from the site of Dikika in Ethiopia (Alemseged et al. 2006). The spectacular skeleton is approximately 3.3 million years old. Such a rare find is great news for paleoanthropologists, since its completeness provides much-needed information about growth and development, juvenile morphology, and even bones that rarely or almost never preserve well from hominins, including a scapula (part of the shoulder) and hyoid bone (sits in the middle of the throat, unwilling to be friends with any other bones). All in all, it’s a very interesting specimen, whose feet show that it was adapted for bipedalism. But its “gorilla-like” scapula may indicate some degree of climbing behavior. The find made the cover of Nature, and here’s part of Figure 1 from the paper:
 Now compare this to Jen, a gelfling from the 1982 Jim Henson film The Dark Crystal.
Now compare this to Jen, a gelfling from the 1982 Jim Henson film The Dark Crystal.
 Creepy. But the resemblance is dead-on, just look at the prognathic faces of DIK-1-1, above, and Jen here.
Creepy. But the resemblance is dead-on, just look at the prognathic faces of DIK-1-1, above, and Jen here.
So what do we learn? Most probably A. afarensis is ancestral to the gelflings, as well as later, more well-known hominins like A. africanus, robustus, boisei, and our genus, Homo. I suppose the gelflings were an evolutionary ‘side-branch.’ And since DIK-1-1 is a juvenile while this gelfling is an adult, we have documented here a case of paedomorphosis, an evolutionary phenomenon in which the adults of the descendant taxon appear more similar to the juveniles of their ancestors (for a real-life example of this, see the axolotl).
Also, Alemseged et al. posit that the gorilla-like morphology of the Dikika scapula may reflect climbing behavior. Well, if we remember The Dark Crystal, we’ll recall that Jen climbed Aughra‘s model solar system with gusto when the bad guys came and messed the place up. So the functional interpretation of the fossil shoulder is corroborated with behavioral data from the animatronic puppet. Oh, also I think the gelflings lived in a wooded, perhaps even forest environment. Such environments likely characterized the habitats of earlier hominins, but isotopic and relative abundances of different kinds of other fossil animals suggest that Dikika may have been a bit more open (Wynn et al. 2006).
![]() References
References
Alemseged Z, Spoor F, Kimbel WH, Bobe R, Geraads D, Reed D, & Wynn JG (2006). A juvenile early hominin skeleton from Dikika, Ethiopia. Nature, 443 (7109), 296-301 PMID: 16988704
Wynn J, Alemseged Z, Bobe R, Geraads D, Reed D, and Roman D. 2006. Geological and paleontological context of a Pliocene juvenile hominin at Dikika, Ethiopia. Nature 443: 332-336.
*Edited 08 Nov 2015
Long-term effects of hybridization in primates
One of my research interests is hybridization in primates, and the possible role it played in hominin evolution. It’s a sticky subject, so it’s always fun to find good papers on real-life examples of hybridization between different primate ‘species.’ In this vein, Burrell et al. <!–[if supportFields]> ADDIN EN.CITE Burrell200975375317Burrell, Andrew S.Jolly, Clifford J.Tosi, Anthony J.Disotell, Todd R.Mitochondrial evidence for the hybrid origin of the kipunji, Rungwecebus kipunji (Primates: Papionini)Molecular Phylogenetics and EvolutionMolecular Phylogenetics and Evolution340-348512KipunjiBaboonRungwecebusLophocebusPapioHybrid speciationMangabey2009http://www.sciencedirect.com/science/article/B6WNH-4VNKGV7-4/2/1fc25562a43afdf7c3fafda3bcfaceb7 <![endif]–>(2009)<!–[if supportFields]><![endif]–> report that the kipunji—a highly endangered papionin monkey from a small area in Tanzania—has an mtDNA haplotype from its yellow baboon (Papio cynocephalus) neighbors. Morphologically, the (living) monkey looks more like mangabeys (Lophocebus), though it has some baboon-like affinities, too.The authors posit that the most likely reason for this is inter-generic hybridization in the past, between Papio cynocephalus (yellow baboons) and Lophocebus sp. (mangabey monkeys).
The authors suggest a scenario in which in a marginal environment, Lophocebus (or Cercocebus?) males mated with some P. cynocephalus females. The hybrids, then, back-crossed into the respective parent species—thus baboon mtDNA was brought into a mangabey population. From here, the habitat favored nuclear genes of mangabeys, hence the overall mangabey appearance. Even though mtDNA is often (by necessity) assumed to be selectively neutral for phylogenetic studies such as these, it is not inconceivable that the baboon mtDNA persisted in the population because of selection, too.
The authors note that the test of the hybrid-origin hypothesis will come from nuclear DNA. If the kipunji truly represents the meshing of two genera’s genomes, then it should have a large amount of mangabey nuclear DNA. However, if the nuclear genome is all Papio that would mean that the kipunji’s ancestors were baboons whose morphology (and niche?) converged on that of mangabeys. Even this outcome would be a bit incredible, given the apparent pervasiveness of homoplasy within the papionins. In fact, the few nuclear genes known for the specimen either cluster in Papio, or are phylogenetically ambiguous. But for the moment, mtDNA and morphology support hybrid-origins. This is especially remarkable, since hybridization between genera, above the species level, leading to a stable taxon has not been documented before.
It is unclear whether the kipunji represents an instance of hybrid (or “secondary”) speciation, in which hybrids thrive in an environment while individuals of the parental species don’t, or just an intense case of gene transfer between species (I suppose if you’re getting a whole mitochondrial genome, it’s not really introgression). Nevertheless, the paper provides an amazing example of the potential evolutionary significance of hybridization in primates. Nice.
References
<!–[if supportFields]> ADDIN EN.REFLIST <![endif]–>Burrell AS, Jolly CJ, Tosi AJ, and Disotell TR. 2009. Mitochondrial evidence for the hybrid origin of the kipunji, Rungwecebus kipunji (Primates: Papionini). Molecular Phylogenetics and Evolution 51(2):340-348.
A New African Late Miocene Ape
Martin Pickford and colleagues recently announced the discovery of a fossil ape from Niger. Fauna from the site suggest an age of anywhere from 11 – 5 million years ago. The fossil is just a fragment of the right mandible, containing the roots of the first molar. The form of the molar roots show its affinities with hominoid primates. Aside from that, little can be said about the fragment. It is very slender, unlike any hominin. The authors note that the size of the jaw and aspects of the root ally it closely with modern chimpanzees, but it does not preserve any diagnostic features that link it specifically to any living or fossil ape.
This is an exciting new find that addresses two big questions in paleoanthropology. First, the African late Miocene has a poor hominoid fossil record. Exceptions to this are the potential hominins Sahelanthropus, Orrorin, and Ardipithecus kadabba; and the ape-like Chororapithecus, Nakalipithecus, and Samburupithecus. So this new specimen, whether it represents one of the already-known fossil apes or is a new taxon, provides further evidence that apes were present in Africa in this time period. This bears on the debate about whether the living African apes (gorillas and chimpanzees/bonobos) originated in Eurasia or Africa, though certainly more fossils are needed to address this.
Second, most African fossil hominoids are known only from Eastern Africa. As such, it has looked like much of hominoid and hominin evolution have taken place there. At the moment, the only other non-East-African fossil hominoids I can think of are the 13 million year old Otavipithecus mandible from Namibia, and the 7 million year old Sahelanthropus fossils from Chad. So it is clear that hominoids (and hominins) existed in places other than East and South Africa. As the authors note, it is not unlikely that more hominoids (and hominins?) will be discovered in western and central Africa. Who knows, we might even get some (more?) chimpanzee ancestors out of it, a shift in the spotlight from our lineage to that of our closest living relatives.
Source:
Pickford M, Coppens Y, Senut B, Morales J, and Braga J. Late Miocene hominoid from Niger. Comptes Rendus Palevol In Press, Corrected Proof.
Guest Post: Jerry and Julie on their latest paper
“Good news, everyone!” to quote Prof. Farnsworth. Our good friends Jerry DeSilva and Julie Lesnik just published a paper in the Journal of Human Evolution, about neonatal brain size in primates [1]. Rather than talk and talk about it, probably missing the important stuff, I made some calls. The authors were kind enough to make a cameo appearance at Lawn Chair to talk about their paper about their paper. Thanks, Jerry and Julie! Here’s what they had to say:
Summary:
This paper presents a regression equation that can be used to calculate the size of the brain at birth in different hominin species.
Significance:
Knowing the size of the brain at birth is critical for understanding obstetric constraints and brain development throughout human evolution. Unfortunately, it is very unlikely to find fossil evidence of how big the brain was at birth in human ancestors (though see below). This paper presents a way to get around the absence of fossil evidence and calculate the size of the neonatal brain in early homs using what we know about brain development in modern primates.
Things Jerry liked about the paper:
Humans are so unusual, and in biological anthropology we often study ways in which humans are different from other primates. However, what this study finds is that humans are no different from other primates in terms of the adult/neonatal brain scaling relationship. This means that we have exactly the brain size at birth expected given the size of our brains as adults. Because of this, we can infer that our extinct ancestors and relatives also followed this ‘rule’ of adult/neonatal brain size, and can calculate the size of the brain at birth from reliable estimates of brain size in 89 adult fossil crania that have been unearthed.
I am also thrilled that Julie and I may have solved the “% brain size at birth” issue that has been all over the literature lately. Did Homo erectus have a more human-like or a more chimpanzee-like pattern of brain growth? What about australopiths? Well, we’ve found that the whole issue of % brain size at birth is simply a function of the scaling relationship between adult and neonatal brain size. Because they do not scale 1:1, but instead scale 1:0.73 (roughly), as the adult brain gets bigger, the neonatal brain gets proportionately smaller. Therefore, less of brain growth occurs in the womb as overall adult brain size increases. If you know the size of a hominin brain as an adult (which we do from the many preserved fossil crania), you can calculate the size of the brain as a baby, and then easily take a % of how much of that brain growth is achieved by birth.
Again, because of the negative allometry (m=0.73), we argue that % of brain size at birth in hominins was never “chimpanzee-like” or “human-like”, but instead followed a gradual progression from a chimpanzee-like ancestral condition to what we have today.
Things Julie liked about the paper:
So much is going on when we think about hominid evolution, especially in the early Pleistocene. With the emergence of Homo brain size is increasing, bipedality is becoming more efficient, and tool use is becoming more advanced. What I like about this paper is that understanding neonatal brain size is one way of tying all of those elements together. Humans are considered to be secondarily altricial meaning that they are born in a more underdeveloped state than their ancestors. Selection for this smaller neonatal size is often considered to be linked to the constraints placed on the pelvis by selection for more efficient bipedal locomotion. A small brain size at birth and a large adult brain always seemed exceptional for Homo. What our paper shows is that the relationship is entirely normal across anthropoids. So, where is the selective pressure? On the larger brain as an adult or on the smaller brain as a newborn? I am now more apt to lean towards larger adult brain. Efficient bipedality is important for exactly that reason; it’s efficient and therefore requires less energy to walk upright and allows the body to allot that energy to other tasks, such as maintenance of a large brain. Add tool-use advancement to the equation and it seems bigger brains and more advanced cognitive abilities were of primary importance at this stage of human evolution.
What we’d do different:
I would have included Neandertals. Julie and I made a statement in the introduction that the discovery of neonatal crania was bordering on impossible. Just days before our paper appeared on-line, however, Marcia Ponce de Leon published a fantastic paper in PNAS on a neonatal Neandertal cranium from Mezmaiskaya Cave in Russia [2]. What is very exciting to me is that this newly described fossil allows us to test our regression equation. How accurate is it in predicting the size of the brain at birth in Neandertals (which we now know because of this new specimen)? Our regression would predict a brain size of about 425 cc, which is very close to the size of the brain at birth in the Mezmaiskaya infant and well within the 95% CI. When two independent methods arrive at the same result, it is reasonable to argue that the method is valid.
Referenecs
1. DeSilva J, and Lesnik J. 2008. Brain size at birth throughout human evolution: a new method for estimating neonatal brain size in hominins. Journal of Human Evolution, corrected proof in press.
2. Ponce de Leon M, Golovanova L, Doronichev V, Ramanova G, Akazawa T, Kondo O, Ishima H, and Zollikofer C. 2008. Neanderthal brain size at birth provides insights into the evolution of human life history. Proceedings of the National Academy of Sciences 105: 13764-13768
New twist from teeth
Peter Ungar, Fred Grine and Mark Teaford recently reported in PLoS ONE on their results of studying the microwear on Australopithecus boisei molars. Their study showed that the microwear differs from that of A. robustus, arguably boisei‘s South African counterpart, and from A. africanus. Here’s the abstract:
The Plio-Pleistocene hominin Paranthropus boisei had enormous, flat, thickly enameled cheek teeth, a robust cranium and mandible, and inferred massive, powerful chewing muscles. This specialized morphology, which earned P. boisei the nickname “Nutcracker Man”, suggests that this hominin could have consumed very mechanically challenging foods. It has been recently argued, however, that specialized hominin morphology may indicate adaptations for the consumption of occasional fallback foods rather than preferred resources. Dental microwear offers a potential means by which to test this hypothesis in that it reflects actual use rather than genetic adaptation. High microwear surface texture complexity and anisotropy in extant primates can be associated with the consumption of exceptionally hard and tough foods respectively. Here we present the first quantitative analysis of dental microwear for P. boisei. Seven specimens examined preserved unobscured antemortem molar microwear. These all show relatively low complexity and anisotropy values. This suggests that none of the individuals consumed especially hard or tough foods in the days before they died. The apparent discrepancy between microwear and functional anatomy is consistent with the idea that P. boisei presents a hominin example of Liem’s Paradox, wherein a highly derived morphology need not reflect a specialized diet.
References
Ungar PS, Grine FE, and Teaford MF. 2008. Dental Microwear and Diet of the Plio-Pleistocene Hominin Paranthropus boisei. PLoS ONE 3(4):e2044.
Wood B, and Constantino P. 2007. Paranthropus boisei: Fifty years of evidence and analysis. American Journal of Physical Anthropology 134(S45):106-132.