David Krause and colleagues announced in this week’s Nature the discovery of a new species of extinct mammal, Vintana sertichi, that lived in what is now Madagascar between 66-72 million years ago. The species is based on a very well-preserved cranium of an early gondwanatherian (if you want to impress your friends this weekend, gratuitously use the word “gondwanatherian”). I don’t know much about early mammals like this, but it sounds like it was a weird creature (see the Stony Brook press release). Just looking at it’s face there’s something that sticks out as strange:

Vintana sertichi cranium (Reich et al. 2014, Figure 1a). On the left is a 3D CT reconstruction, and on the right is a line drawing highlighting all the individual bones (so many cranial bones). The view is from the right side, so the nose is to the right, the eye socket is the shadowy hollow in the middle, and the back of the skull is on the left. The jugal flanges are the downward projections.
Jutting downward from the sides of the jaw are ‘jugal flanges,’ projections of bone on the homologs of human cheeks. Projections of like these usually serve as muscle attachment sites, and the size of the projection generally reflects the size of the muscle. These facial flanges anchor the masseter muscle, a major chewing muscle that helps close the jaw. The size of this flange in Vintana suggests its chomp packed a punch. A debilitating bite. A face not even a mother could love (so now they’re extinct).
Vintana‘s bony tear-catchers caught my eye because most primates I’ve seen have, you know, less heinous faces. Scouring the internet, big jugal flanges are a fairly rare sight, but can apparently be found in glyptodonts (giant, armadillo-like mammals that lived tens of thousands of years ago) and various sloths. The closest thing I’ve seen to this gross bony flange in Primates are on the zygomatic bones of some extinct, baboon-like animals, such as Dinopithecus ingens:
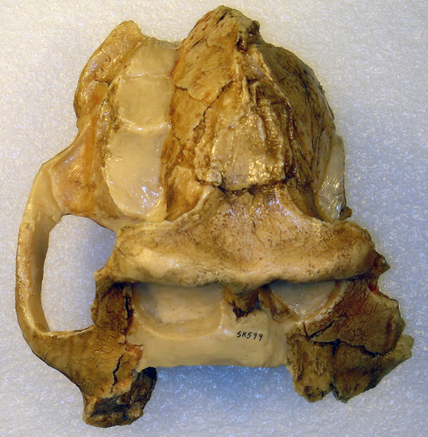
Fragmentary skull, viewed from the top, of Papio (a.k.a. Dinopithecus) ingens, from Swartkrans, South Africa. As a punishment for its zygomatic excess, its face was confiscated. Photo credit: CalPhotos.
and Theropithecus brumpti

Theropithecus brumpti from the Omo Basin, Ethiopia. Photo credit: CalPhotos.
So some primates dabbled in jugal flangery like Vintana, but Natural Selection was having none of it. Anyway, Vintana overcame this craniofacial adversity with characteristic Mesozoic moxie, and is an important piece in the puzzle of mammal evolution. It will be interesting to see what other mammalian surprises the Mesozoic has in store for paleontologists.


