Two anthropology papers came out yesterday in advance print at the Proceedings of the National Academy of Sciences. I’d like first to draw your attention to the fact that they’re open access – normally such scientific papers are behind a paywall, but these two can be obtained by anyone (well, anyone with internet). One is about the chronology and nature of Acheulean technology at the 1.7-1.0 mya site of Konso in Ethiopia. The other, and the subject of this post, is about life history in wild chimpanzees from Uganda.
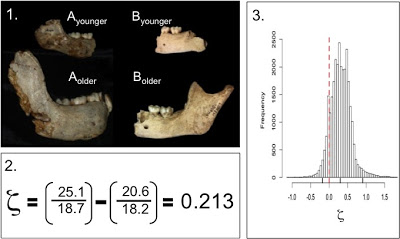
 Tanya Smith and colleagues analyzed behavior of chimps and photographs of chimps’ erupting first molars (“M1”) to determine a] the age at which these events happen in the wild (in this population at least), and b] whether M1 eruption is tightly linked with other important life history variables, such as the adoption of adult foods, as had previously been claimed. What an adorable study – check out figure 1 from the paper (right):
Tanya Smith and colleagues analyzed behavior of chimps and photographs of chimps’ erupting first molars (“M1”) to determine a] the age at which these events happen in the wild (in this population at least), and b] whether M1 eruption is tightly linked with other important life history variables, such as the adoption of adult foods, as had previously been claimed. What an adorable study – check out figure 1 from the paper (right):
Figuring out age at M1 eruption in wild, healthy chimps is important because there has been debate about whether wild chimps actually erupt their teeth at as young of ages as they do in captivity – not natural conditions. This question has recently been investigated in a skeletal sample of wild chimps of known age, from Tai forest in Cote d’Ivoire (Zihlman et al. 2004, T Smith et al. 2010), but somehow these studies raised more questions than they answered (e.g. BH Smith and Boesch 2011). So TM Smith and colleagues decided to further address this question with photographic evidence of living, arguably healthy chimps.
They found that M1 eruption occurred anywhere from 2.8-3.3 years of age in their sample of 5 cuddly infants, consistent with estimates from captivity. I have to say I’m a bit surprised it wasn’t later (but what fun is science if it’s not surprising?). Of course, this is based on 5 infants from one population, so it could stand to be reinvestigated in other chimp populations as the authors note.
Smith et al’s second task was to see how well age at M1 eruption coincided with other life history variables – this is supposed to be an important event, alleged to coincide with cessation of weaning and the adoption of adult foods. Moreover, since a mother is no longer nursing her infant, M1 eruption “should” also be roughly contemporaneous with a mother’s return to estrus cycling and subsequent reproduction. Many infants were observed to begin eating adult-like foods prior to M1 eruption, around 3 years. Unexpectedly however, infants also nursed for a while even after M1 eruption. In fact, time spent nursing actually increased for a brief period around 3 years of age, possibly because their mothers’ milk was not as nutritious as at younger ages.
Now, what interests me most about this are possible implications for the evolution of growth and life history. Many researchers have argued that extinct hominids, like the australopithecines, would have grown up relatively rapidly like apes, rather than slowly like humans. This claim has been based pretty much entirely on dental development, until my dissertation research. There, I’ve shown that one hominid, Australopithecus robustus, probably experienced greater jaw growth than humans prior to eruption of the M2. Now, if this hominid erupted its teeth as fast as apes, and grew more than humans, this implies really really high growth rates for A. robustus (that is, if we can extrapolate from the jaw to the overall body size).
![]() I’ll be working a bit more on this latter point in the near future. In the mean time, let’s hear it for open access bioanthro Continue reading
I’ll be working a bit more on this latter point in the near future. In the mean time, let’s hear it for open access bioanthro Continue reading