Last November I reported on recently described Australopithecus cf. afarensis craniodental remains from the site of Woranso Mille in Ethiopia. These fossils are significant in part because they date to around 3.6 million years ago; most of the postcranial evidence for A. afarensis comes from Hadar (~3.4 – 2.9 million years) or Maka (~3.5 million years). It is pretty awesome, then, that Yohannes Haile-Selassie and colleagues (2010a) have just reported on a partial skeleton from Woranso-Mille.
Last November I reported on recently described Australopithecus cf. afarensis craniodental remains from the site of Woranso Mille in Ethiopia. These fossils are significant in part because they date to around 3.6 million years ago; most of the postcranial evidence for A. afarensis comes from Hadar (~3.4 – 2.9 million years) or Maka (~3.5 million years). It is pretty awesome, then, that Yohannes Haile-Selassie and colleagues (2010a) have just reported on a partial skeleton from Woranso-Mille.
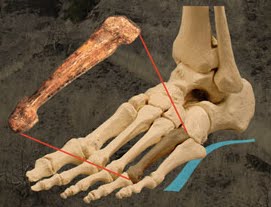
The specimen is given the catalog number KSD-VP-1/1 (right, from Nature), and the nickname Kadanuumuu, meaning “Big Man” in the language of the Afar people who live in the region where the fossils were discovered. Here, I’ll be focusing on the scapula.
Researchers have debated about what the scapular form of A. afarensis means functionally – how could, and did, these creatures use their shoulders? The scapula of AL 288 (the famous “Lucy”) preserves part of the glenoid fossa (shoulder socket) and only a little of the surrounding bone including the scapular spine (below). It has been argued that the angle between the glenoid fossa and the lateral border is more similar to modern apes than to humans. That is, the shoulder socket may have been oriented more upward, like in modern apes, compared to humans whose socket faces more to the side. The implication is that A. afarensis may have been preferentially exploiting arboreal environments.

Left: AL 288 scapular fragment. The glenoid fossa is the hollow that faces to the right, the lateral border is at the bottom paralleling the label “AL 288-1L.” The scapular spine is preserved only at the base, it is the small uprising of bone just to the left of the glenoid fossa. From Haile-Selassie et al. 2010b, Fig. S21.
Similarly, a juvenile afarensis skeleton from the Ethiopian site of Dikika (Alemseged et al. 2006), dating to around 3.4 million years ago, also suggested an ape-like shoulder for this extinct human ancestor. Principal components analysis of several measurements from the Dikika scapula showed it to be very similar to gorillas of comparable age, in terms of overall shape and proportions.
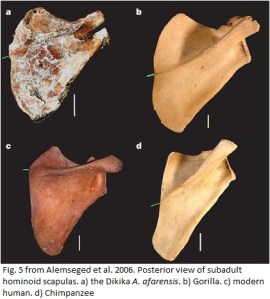
So from these two scapulae, one belonging to a very small-bodied female, the other from a small ~3-year-old possible female, we get the picture that A. afarensis had a fairly ape-like (i.e. arboreal) shoulder orientation, and may not have had independent movement of the head and trunk that we modern humans enjoy. Nevertheless, it is still unclear whether this means that the afarensis scapula functioned like that of an ape, and hence its shape, or whether the similarity in shape is a ‘hold-over’ from having an arboreal ancestor. I will say, I think one very telling feature noticeable in even the fragmentary AL 288 is the relative position and orientation of the scapular spine. Note that in the apes (the two juveniles scapulae on the right of the diagram to the left), the scapular spine roughly parallels the lateral border, and as a result, the flat areas above and below the spine are roughly equal in size. The above area houses the supraspinatus muscle, a rotator cuff muscle that acts largely in elevating the arm above the head and stabilizing the shoulder joint. In humans and afarensis, in contrast, the lower (insfraspinous) fossa is fairly large compared to the upper (supraspinous) fossa. Thus, the argument can be made that in humans and hominids, less power is needed to raise the arms over the head, or that humans and hominids have a greater reliance on the infraspinatus muscle for bringing the arm down toward the body and stabilizing the shoulder joint.

Now, KSD-VP-1 provides a remarkably complete scapula of an adult afarensis (right). In contrast to the specimens described above, KSD-VP-1 is very human-like. To the naked eye, and as borne out by principal components analysis of scapular angles, this thing is very human-like.
Now the question is, why does the morphology of this new specimen seem at odds with Lucy and Dikika? Part of the answer could be scaling – indeed, the authors note that the orientation of the glenoid relative to the lateral border (more specifically the scapular bar) in AL 288 can be found in modern humans of small size.
But that still does not answer the question of why the complete adult afarensis scapula is like adult humans, whereas the child afarensis is like young gorillas. The authors posit that perhaps it is due to Dikika’s fairly large supraspinous fossa. They also suggest that the measurements used in Alemseged et al’s study could not capture functional and discriminatory information about scapula shape. Nevertheless, a simple visual comparison the Dikika and KSD (x-ray…) scapulae reveals them to look fairly different, i.e. Dikika is relatively broader side-to-side.
Could ontogeny explain the differences between the child and adult afarensis? In a study of scapular growth and development in living primates, Young (2008) found childhood growth does not appear to explain adult shape variation. That is to say, most aspects of species-specific morphology are present in subadult scapulae. Rather, most variation in scapular shape among modern primates appears to be due to functional differences: climbers’ scapulae differ consistently from quadrupeds’. So what does that imply? That at 3.59 million years, adult male A. afarensis were not using their shoulders for arboreal activities, but at 3.4 million years ago, subadults were? Maybe this is just normal intraspecific variation? Maybe the ontogeny of the scapulae needs to be examined further?
I have to say I agree with Haile-Selassie et al. (2010a) here, that differences in the statistical analyses between the current study and that of Alemseged et al. (2006) may be partly responsible for the different interpretations of A. afarensis scapular morphology. Still, visual inspection of pictures of the fossils suggests to me that even if the principal components analyses were carried out using the same variables (Alemseged et al. used linear measurements, H-S et al. used angles), Dikika might seem gorilla-like, KSD still human-like; Nota bene that principal components analysis is not actually a test in itself, but rather an exploratory statistical technique. As such, it will never really “tell” how a bone was used. Still, I think this does raise an important issue about scapular function and ontogeny in hominoids.
References
Alemseged Z, Spoor F, Kimble WH, Bobe R, Geraads D, Reed D, and Wynn JG. 2006. A juvenile early hominin skeleton from Dikika, Ethiopia. Nature 443: 296-301.
Haile-Selassie Y, Latimer BM, Alene M, Deino AL, Gibert L, Melillo, Saylor BZ, Scott GR, and Lovejoy CO. 2010a. An early Australopithecus afarensis postcranium from Woranso-Mille, Ethiopia. Proceedings of the National Academy of Sciences, USA, in press.
Haile-Selassie et al. 2010b. Supplementary Online Material to 2010a.
Young NM. 2008. A Comparison of the Ontogeny of Shape Variation in the Anthropoid Scapula: Functional and Phylogenetic Signal. American Journal of Physical Anthropology 136: 247-264.